Post some cool materials. Pic related is aerogel. An ultra light material derived from gel that has had the liquid component replaced with gas.
/cyb/ - Cyberpunk Fiction and Fact
Cyberpunk is the idea that technology will condemn us to a future of totalitarian nightmares here you can discuss recent events and how technology has been used to facilitate greater control by the elites, or works of fiction
File (): E6089F9B50C4A255F20D05A183AAE99F-110427.jpg (107.8 KB, 879x619, Aerogel_hand (1).jpg)

Anonymous
No.918
File (): 0151415210DAF97BCEBC9EAA079BB902-1783697.jpg (1.7 MB, 2000x1500, Main-1.jpg)

Damascus Steel. An ancient steel alloy which was found to have carbon nano tubes in it.
Anonymous
No.922
File (): 7E2E82646108143071522264FE55DA1F-19951.jpg (19.5 KB, 220x293, 220px-Aluminium_foam.jpg)

A metal foam is a cellular structure consisting of a solid metal (frequently aluminium) with gas-filled pores comprising a large portion of the volume. The pores can be sealed (closed-cell foam) or interconnected (open-cell foam). The defining characteristic of metal foams is a high porosity: typically only 5–25% of the volume is the base metal, making these ultralight materials. The strength of the material is due to the square-cube law.
Metallic foams typically retain some physical properties of their base material. Foam made from non-flammable metal remains non-flammable and can generally be recycled as the base material. Its coefficient of thermal expansion is similar while thermal conductivity is likely reduced.[1]
Metallic foams typically retain some physical properties of their base material. Foam made from non-flammable metal remains non-flammable and can generally be recycled as the base material. Its coefficient of thermal expansion is similar while thermal conductivity is likely reduced.[1]
Anonymous
No.927
File (): 25CF48BF6F5273AB90A191EA2DD0F7E7-36537.jpg (35.7 KB, 560x353, amorphous.metal.jpg)

Amorphous metals, also called metallic glasses, consist of metal with a disordered atomic structure. They can be twice as strong as steel. Because of their disordered structure, they can disperse impact energy more effectively than a metal crystal, which has points of weakness. Amorphous metals are made by quickly cooling molten metal before it has a chance to align itself in a crystal pattern. Amorphous metals may be the military’s next generation of armor, before they adopt diamondoid armor in mid-century. On the green side of things, amorphous metals have electronic properties that improve the efficiency of power grids by as much as 40%, saving us thousands of tons of fossil fuel emissions.
Anonymous
No.934
File (): 023C2F23FAF26C6C544C0857C0BE2DBE-802268.jpg (783.5 KB, 1280x1024, Graphen.jpg)

Graphene is a single layer (monolayer) of carbon atoms, tightly bound in a hexagonal honeycomb lattice. It is an allotrope of carbon in the form of a plane of sp2-bonded atoms with a molecular bond length of 0.142 nanometres. Layers of graphene stacked on top of each other form graphite, with an interplanar spacing of 0.335 nanometres. The separate layers of graphene in graphite are held together by van der Waals forces, which can be overcome during exfoliation of graphene from graphite.
Graphene is the thinnest compound known to man at one atom thick, the lightest material known (with 1 square meter weighing around 0.77 milligrams), the strongest compound discovered (between 100-300 times stronger than steel with a tensile strength of 130 GPa and a Young's modulus of 1 TPa - 150,000,000 psi), the best conductor of heat at room temperature (at (4.84±0.44) × 10^3 to (5.30±0.48) × 10^3 W·m−1·K−1) and also the best conductor of electricity known (studies have shown electron mobility at values of more than 200,000 cm2·V−1·s−1). Other notable properties of graphene are its uniform absorption of light across the visible and near-infrared parts of the spectrum (πα ≈ 2.3%), and its potential suitability for use in spin transport.
Bearing this in mind, one might be surprised to know that carbon is the second most abundant mass within the human body and the fourth most abundant element in the universe (by mass), after hydrogen, helium and oxygen. This makes carbon the chemical basis for all known life on earth, making graphene potentially an eco-friendly, sustainable solution for an almost limitless number of applications. Since the discovery (or more accurately, the mechanical obtainment) of graphene, applications within different scientific disciplines have exploded, with huge gains being made particularly in high-frequency electronics, bio, chemical and magnetic sensors, ultra-wide bandwidth photodetectors, and energy storage and generation.
Graphene is the thinnest compound known to man at one atom thick, the lightest material known (with 1 square meter weighing around 0.77 milligrams), the strongest compound discovered (between 100-300 times stronger than steel with a tensile strength of 130 GPa and a Young's modulus of 1 TPa - 150,000,000 psi), the best conductor of heat at room temperature (at (4.84±0.44) × 10^3 to (5.30±0.48) × 10^3 W·m−1·K−1) and also the best conductor of electricity known (studies have shown electron mobility at values of more than 200,000 cm2·V−1·s−1). Other notable properties of graphene are its uniform absorption of light across the visible and near-infrared parts of the spectrum (πα ≈ 2.3%), and its potential suitability for use in spin transport.
Bearing this in mind, one might be surprised to know that carbon is the second most abundant mass within the human body and the fourth most abundant element in the universe (by mass), after hydrogen, helium and oxygen. This makes carbon the chemical basis for all known life on earth, making graphene potentially an eco-friendly, sustainable solution for an almost limitless number of applications. Since the discovery (or more accurately, the mechanical obtainment) of graphene, applications within different scientific disciplines have exploded, with huge gains being made particularly in high-frequency electronics, bio, chemical and magnetic sensors, ultra-wide bandwidth photodetectors, and energy storage and generation.
Anonymous
No.939
Anonymous
No.950
File (): 07258880D4EC67D19374B593392C1874-66149.jpg (64.6 KB, 1200x675, serveimage (1).jpg)

>Rayon is a manufactured fiber made from regenerated cellulose fiber. The many types and grades of rayon can imitate the feel and texture of natural fibers such as silk, wool, cotton, and linen. The types that resemble silk are often called artificial silk.
Anonymous
No.955
File (): 99A00C18807FA865E4B4C4C39323D8CA-73725.jpg (72.0 KB, 1024x1024, FKT5342IWWFJ13K.LARGE.jpg)

Casein plastics were introduced at the beginning of the 20th century, their starting material being the protein in cows milk, precipitated by the action of the enzyme rennin.
Although casein is readily moulded to shape under moderate heat and pressure, it does not produce a stable material for manufacture until it has become hardened by soaking in formalin (5% solution of formaldehyde in water) for a long period. Unfortunately, this causes much distortion so casein plastics are almost always produced by machining stock material such as sheet, rod, tube or buttton blanks (small discs). After machining, casein may be polished either mechanically with abrasives or chemically with a ‘dip polish’.
The material readily takes a surface dye, so coloured items can be quickly made from pale coloured stock items. This was especially important for the button trade which was the principal consumer of casein plastics.
As well as buttons and buckles, casein was also used for knitting pins, fountain pen and propelling pencil barrels, dressing table ware and a host of other items.
Although casein is readily moulded to shape under moderate heat and pressure, it does not produce a stable material for manufacture until it has become hardened by soaking in formalin (5% solution of formaldehyde in water) for a long period. Unfortunately, this causes much distortion so casein plastics are almost always produced by machining stock material such as sheet, rod, tube or buttton blanks (small discs). After machining, casein may be polished either mechanically with abrasives or chemically with a ‘dip polish’.
The material readily takes a surface dye, so coloured items can be quickly made from pale coloured stock items. This was especially important for the button trade which was the principal consumer of casein plastics.
As well as buttons and buckles, casein was also used for knitting pins, fountain pen and propelling pencil barrels, dressing table ware and a host of other items.
Anonymous
No.958
File (): 5F80FB5D5E01A13B803F16C201DEA341-126985.png (124.0 KB, 1024x1006, Buckminsterfullerene.svg.png)
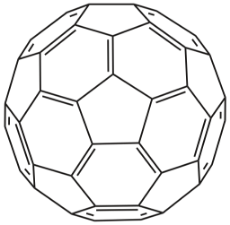
Buckminsterfullerene is a type of fullerene with the formula C60. It has a cage-like fused-ring structure (truncated icosahedron) that resembles a soccer ball (football), made of twenty hexagons and twelve pentagons, with a carbon atom which has one π bond and two single bonds at each vertex of each polygon and a bond along each polygon edge. These have a lot of potential applications in medicine and solar cells.
Anonymous
No.962
File (): 515162D2336589DF887E63F7CC57251E-31288.jpg (30.6 KB, 480x480, Multi-Walled-Carbon-Nanotube-Structure-lc_grande.jpg)
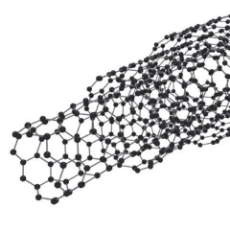
Carbon nanotubes are long chains of carbon held together with sp2 bonds that are stronger than the sp3 bonds in diamonds! These remarkable structures have countless amazing properties. These include ballistic electron transport, great for electronics, as very high tensile strength making them a candidate for potential applications such as space elevators.
Carbon nanotubes have specific strengths of up to 48,000 kN.m/kg, stronger than most other materials known. High carbon steel, by way of example, is 154 kN.m/kg. Nanotubes are therefore around 300 times stronger than steel! You could build incredibly tall towers, perhaps kilometers high, with a material like this.
Carbon nanotubes have specific strengths of up to 48,000 kN.m/kg, stronger than most other materials known. High carbon steel, by way of example, is 154 kN.m/kg. Nanotubes are therefore around 300 times stronger than steel! You could build incredibly tall towers, perhaps kilometers high, with a material like this.
Anonymous
No.968
File (): AD54D237E17B09D8DA5B8DE11F8C718C-5921.jpg (5.8 KB, 220x147, Bronze_bell_with_visible_material_structure.jpg)

Bronze is an alloy consisting primarily of copper, commonly with about 12–12.5% tin and often with the addition of other metals (such as aluminium, manganese, nickel or zinc) and sometimes non-metals or metalloids such as arsenic, phosphorus or silicon. These additions produce a range of alloys that may be harder than copper alone, or have other useful properties, such as stiffness, ductility, or machinability.
Anonymous
No.977
File (): C847D4F511C256B4928F0DBA95840245-16869.jpg (16.5 KB, 220x165, 220px-Milyanfan-adobe-bricks-8038.jpg)

Adobe is a building material made from earth and organic materials. Adobe is Spanish for mudbrick, but in some English-speaking regions of Spanish heritage, the term is used to refer to any kind of earth construction. Most adobe buildings are similar in appearance to cob and rammed earth buildings. Adobe is among the earliest building materials, and is used throughout the world.
Anonymous
No.982
File (): 665C7D90DF8484BED3331A6DE07E635A-126530.jpg (123.6 KB, 800x533, Hempcrete-blocks.jpg)

Hempcrete or Hemplime is bio-composite material, a mixture of hemp hurds and lime which is used as a material for construction and insulation. It is marketed under names like Hempcrete, Canobiote, Canosmose, and Isochanvre.
File (): BD6B53BA326B031F718E94F17B8D17C3-713.gif (713.0 bytes, 124x96, Tetraethylmethane.gif)
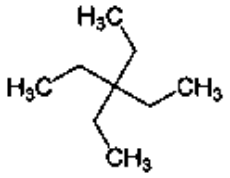
Anonymous
No.989
File (): 989B65354DDAB14FE904B03860FD1AC0-185995.png (181.6 KB, 1024x1104, 1024px-Orthocarbonic-acid-Spartan-MP2-3D-balls-B.png)
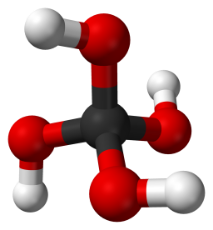
Anonymous
No.992
File (): 0230A02725AC39FCCD3E15484BB2BEAD-57460.jpg (56.1 KB, 630x211, silicene_Fig1a.jpg)

Silicene is a two-dimensional allotrope of silicon, with a hexagonal honeycomb structure similar to that of graphene. Contrary to graphene, silicene is not flat, but has a periodically buckled topology; the coupling between layers in silicene is much stronger than in multilayered graphene; and the oxidized form of silicene, 2D silica, has a very different chemical structure from graphene oxide.
Anonymous
No.1003
File (): 66BB197D48681156EE54AA1A98D40E22-18199.jpg (17.8 KB, 259x194, download (31).jpeg)
Pyrite nanoparticles can be used for more efficient batteries or for solar panels.
Anonymous
No.1011
File (): 73EEF5D26F4B7C2EAA59A6390F101289-15923.jpg (15.5 KB, 275x183, spidersilk.jpg)

Spider silk is a protein fibre spun by spiders. Spiders use their silk to make webs or other structures, which function as sticky nets to catch other animals, or as nests or cocoons to protect their offspring, or to wrap up prey. They can also use their silk to suspend themselves, to float through the air, or to glide away from predators. Most spiders vary the thickness and stickiness of their silk for different uses.
Anonymous
No.1017
File (): C3B581195EAF665BD022041A3455279B-7154.jpg (7.0 KB, 184x274, download (1).jpg)

Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. At room temperature and pressure, another solid form of carbon known as graphite is the chemically stable form, but diamond almost never converts to it. Diamond has the highest hardness and thermal conductivity of any natural material, properties that are utilized in major industrial applications such as cutting and polishing tools. They are also the reason that diamond anvil cells can subject materials to pressures found deep in the Earth.
Anonymous
No.1022
File (): 2C75EB2E8D8B4E9C047333A1278D5CA3-78747.jpg (76.9 KB, 1060x530, o-PLASTIC-facebook-1060x530.jpg)

Researchers at the Georgia Institute of Technology have developed a material from two naturally-occurring polymers that could offer an environmentally-friendly alternative to traditional plastic packaging for food and consumer goods. The material is made by layering cellulose – the most abundant biopolymer on Earth – with chitin, which is widely found in shellfish, insects and fung
Anonymous
No.1030
File (): 873B8EFF0AADF95793FA6E0156E20079-378211.jpg (369.3 KB, 1200x900, 1200px-Rome-Pantheon-Interieur1.jpg)

Roman concrete, also called opus caementicium, was a material used in construction during the late Roman Republic. Roman concrete was based on a hydraulic-setting cement. Roman concrete is durable due to its incorporation of volcanic ash, which prevents cracks from spreading.
Anonymous
No.1033
File (): 15D08F55C03DEB3681AB420C3EC0C275-8166.jpg (8.0 KB, 276x182, download (32).jpeg)

Tethonite
3D-printed objects always look and feel inferior to objects made with traditional manufacturing processes and materials. Tethonite is different: It’s a 3D-printed ceramic compound that, when fired and cured, looks identical to ceramic made by hand or industrial machines. Not only could Tethonite push the boundaries of the ceramic arts by making increasingly intricate designs possible; it also potentially has wide-ranging consumer applications. “Companies like Apple want to figure out new ways to use ceramics, because it’s such an incredible material,” says Dent. “It’s so hard, subtle, and luminous, but unlike metal, ceramic is also brittle.” It breaks, which is why you don’t see it in many gadgets (although the Apple Watch Edition recently introduced a high-end ceramic model). Tethonite could let companies like Apple intimately combine the best qualities of metal and ceramics to create resilient new devices.
3D-printed objects always look and feel inferior to objects made with traditional manufacturing processes and materials. Tethonite is different: It’s a 3D-printed ceramic compound that, when fired and cured, looks identical to ceramic made by hand or industrial machines. Not only could Tethonite push the boundaries of the ceramic arts by making increasingly intricate designs possible; it also potentially has wide-ranging consumer applications. “Companies like Apple want to figure out new ways to use ceramics, because it’s such an incredible material,” says Dent. “It’s so hard, subtle, and luminous, but unlike metal, ceramic is also brittle.” It breaks, which is why you don’t see it in many gadgets (although the Apple Watch Edition recently introduced a high-end ceramic model). Tethonite could let companies like Apple intimately combine the best qualities of metal and ceramics to create resilient new devices.
Anonymous
No.1038
File (): DB181CCD467FFF0665850C562B2D2C81-1295889.jpg (1.2 MB, 2364x2364, cobbauge-cob-building-dezeen-sq.jpg)

Cob, cobb or clom is a natural building material made from subsoil, water, fibrous organic material, and sometimes lime. The contents of subsoil naturally vary, and if it does not contain the right mixture it can be modified with sand or clay.
A molecular “superglue” based on flesh-eating bacteria
Note to journalists: Please report that this research was presented at a meeting of the American Chemical Society.
NEW ORLEANS, April 11, 2013 — In a classic case of turning an enemy into a friend, scientists have engineered a protein from flesh-eating bacteria to act as a molecular “superglue” that promises to become a disease fighter. And their latest results, which make the technology more versatile, were the topic of a report here today at the 245th National Meeting & Exposition of the American Chemical Society, the world’s largest scientific society.
https://www.acs.org/content/acs/en/pressroom/newsreleases/2013/april/a-molecular-superglue-based-on-flesh-eating-bacteria.html&ved=2ahUKEwjVjKj_t9HlAhVER60KHaSzCGMQFjAAegQIARAB&usg=AOvVaw3jWJeg4FP9zN_Rwm-k4xSi&cshid=1572900831296
Note to journalists: Please report that this research was presented at a meeting of the American Chemical Society.
NEW ORLEANS, April 11, 2013 — In a classic case of turning an enemy into a friend, scientists have engineered a protein from flesh-eating bacteria to act as a molecular “superglue” that promises to become a disease fighter. And their latest results, which make the technology more versatile, were the topic of a report here today at the 245th National Meeting & Exposition of the American Chemical Society, the world’s largest scientific society.
https://www.acs.org/content/acs/en/pressroom/newsreleases/2013/april/a-molecular-superglue-based-on-flesh-eating-bacteria.html&ved=2ahUKEwjVjKj_t9HlAhVER60KHaSzCGMQFjAAegQIARAB&usg=AOvVaw3jWJeg4FP9zN_Rwm-k4xSi&cshid=1572900831296
Anonymous
No.1044
Chinking & Daubing. Never forget what the hoers have done for the Burgers.
https://youtu.be/8oYpQ4qCSj8
A dirt/clay mixed with dried grass, dried horse manure, and dried bark to be smooshed with a larger filler material in between two logs of the temporary log cabin.
https://youtu.be/8oYpQ4qCSj8
A dirt/clay mixed with dried grass, dried horse manure, and dried bark to be smooshed with a larger filler material in between two logs of the temporary log cabin.
Anonymous
No.1045
>>1041
Link is broken, but this one worked for me:
https://www.acs.org/content/acs/en/pressroom/newsreleases/2013/april/a-molecular-superglue-based-on-flesh-eating-bacteria.html
Link is broken, but this one worked for me:
https://www.acs.org/content/acs/en/pressroom/newsreleases/2013/april/a-molecular-superglue-based-on-flesh-eating-bacteria.html
Anonymous
No.1067
File (): 958F7168F17A63EC00E72EE3BBD99AA2-18067.jpg (17.6 KB, 210x413, Individual_ZnO-Si_nanotube.jpg)

Silicon nanotubes are nanoparticles which create a tube-like structure from silicon atoms. As with silicon nanowires, they are technologically important due to their unusual physical properties, which differ fundamentally to those of bulk silicon. First reports on silicon nanotubes appeared around the year 2000.
File (): 5377D022927FC1D751FE0132CAA5279B-166523.jpg (162.6 KB, 630x687, nature_high_entropy_alloys_chart_WEB.jpg)
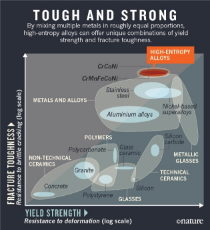
>Roddenberry was right: transparent aluminum exists and is ridiculously strong: 1.6 inches can stop a .50 cal.
>Friction-stir welding, apparently invented in 1991, can join non-ferrous metals without the use of filler
>"High-entropy" alloys are extremely tough and can resist both fracture and deformation like nothing else
>Tantalum and tungsten when doped into titanium create an effective yet lightweight radiation shield
>"Beta cloth" is impossible to cut and has successfully been used in an inflatable module for the ISS
>Composite materials being used in spacesuits are very impact resistant
https://www.extremetech.com/extreme/234925-the-state-of-the-art-in-applied-materials-in-space-explained
>Friction-stir welding, apparently invented in 1991, can join non-ferrous metals without the use of filler
>"High-entropy" alloys are extremely tough and can resist both fracture and deformation like nothing else
>Tantalum and tungsten when doped into titanium create an effective yet lightweight radiation shield
>"Beta cloth" is impossible to cut and has successfully been used in an inflatable module for the ISS
>Composite materials being used in spacesuits are very impact resistant
https://www.extremetech.com/extreme/234925-the-state-of-the-art-in-applied-materials-in-space-explained
Anonymous
No.1085
File (): 9762429A79FC3EC15AAE142BD71D3C43-5960507.mp4 (5.7 MB, Resolution:270x480 Length:00:00:57, Glassblower Skillfully Crafts UV Reactive Sculpture Piece.mp4) [play once] [loop]

Glassblower - Reactive Sculpture Piece